Publications
Holten-Andersen Group Research

Metal Ion Coordinate Polymer Networks
The viscoelastic mechanical properties of hydrogel network can be programmed using bio-inspired metal-coordinate crosslinks sensitive to UV light. Depending on the metal ion used to crosslink the hydrogel, the stiffness can be increased by 1000x, decreased by 100x, or remain unchanged by UV-irradiation.
Grindy, Holten-Andersen, Bio-inspired metal-coordinate hydrogels with programmable viscoelastic material functions controlled by longwave UV light, Soft Matter, 2017.

Polymer-Nanoparticle Composites
Interactions between polymer molecules and inorganic nanoparticles can play a dominant role in nanocomposite material mechanics, yet control of such interfacial interaction dynamics remains a significant challenge particularly in water. This study presents insights on how to engineer hydrogel material mechanics via nanoparticle interface-controlled cross-link dynamics. Inspired by the adhesive chemistry in mussel threads, we have incorporated iron oxide nanoparticles (Fe3O4 NPs) into a catechol-modified polymer network to obtain hydrogels cross-linked via reversible metal-coordination bonds at Fe3O4 NP surfaces. The structurally controlled hierarchical mechanics presented here suggest how to develop hydrogels with remote-controlled self-healing dynamics.
Li, Barrett, Messersmith, Holten-Andersen, Controlling Hydrogel Mechanics via Bio-Inspired Polymer–Nanoparticle Bond Dynamics, ACS Nano, 2015.

Bio-mineralization
Biological organic-inorganic materials remain a popular source of inspiration for bioinspired materials design and engineering. Inspired by the self-assembling metal-reinforced mussel holdfast threads, we tested if metal-coordinate polymer networks can be utilized as simple composite scaffolds for direct in situ crosslink mineralization. Starting with aqueous solutions of polymers end-functionalized with metal-coordinating ligands of catechol or histidine, here we show that inter-molecular metal-ion coordination complexes can serve as mineral nucleation sites, whereby significant mechanical reinforcement is achieved upon nanoscale particle growth directly at the metal-coordinate network crosslink sites.
Kim, Regitsky, Song, Ilavsky, McKinley, and Holten-Andersen, In situ mechanical reinforcement of polymerhydrogels via metal-coordinated crosslink mineralization, Nature Communications, 2021.

Soft Viscoelastic Magnetic Hydrogels
The design of soft magnetic hydrogels with high concentrations of magnetic particles is complicated by weak retention of the iron oxide particles in the hydrogel scaffold. Here, we propose a design strategy that circumvents this problem through the in situ mineralization of iron oxide nanoparticles within polymer hydrogels functionalized with strongly iron-coordinating nitrocatechol groups. The mineralization process facilitates the synthesis of a high concentration of large iron oxide nanoparticles (up to 57 wt % dry mass per single cycle) in a simple one-step process under ambient conditions. The resulting hydrogels are soft (kPa range) and viscoelastic and exhibit strong magnetic actuation. This strategy offers a pathway for the energy-efficient design of soft, mechanically robust, and magneto-responsive hydrogels for biomedical applications.
Song, Kim, Saouaf, Owens, McKinley, and Holten-Andersen, Soft Viscoelastic Magnetic Hydrogels from the In Situ Mineralization of Iron Oxide in Metal-Coordinate Polymer Networks, ACS Appl. Mater. Interfaces, 2023.
Cross-Linking Metal Coordination Gels

The change from wet and soft to dry and hard is a visco-elastic to solid material transition widely displayed in nature, in particular in materials rich in metal-coordinate cross-linking. How metal-coordinate cross-link dynamics contribute to macromolecular material mechanics upon solidification by dehydration remains an open question. Using mussel-inspired Fe-catechol cross-linked polymer hydrogels, we address this question. In addition to a nearly two-fold increase in stiffness, we find that the presence of Fe-catechol coordination bonds in a dehydrated polymer gel also provides the bulk network with a significantly increased energy dissipation with over three times higher loss factor.
Kim, Peterson, Holten-Andersen, Enhanced Water Retention Maintains Energy Dissipation in Dehydrated Metal-Coordinate Polymer Networks: Another Role for Fe-Catechol Cross-Links?, Chemistry of Materials, 2018.
Expanding the stoichiometric window for metal cross-linked gel assembly using competition
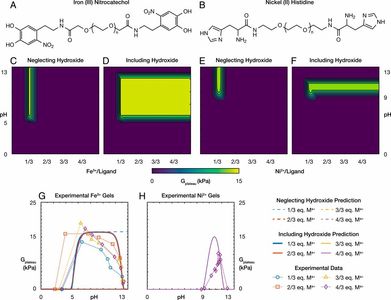
Polymer networks with dynamic cross-links have generated widespread interest as tunable and responsive viscoelastic materials. However, narrow stoichiometric limits in cross-link compositions are typically imposed in the assembly of these materials to prevent excess free cross-linker from dissolving the resulting polymer networks. Here we demonstrate how the presence of molecular competition allows for vast expansion of the previously limited range of cross-linker concentrations that result in robust network assembly. Specifically, we use metal-coordinate cross-linked gels to verify that stoichiometric excessive metal ion cross-linker concentrations can still result in robust gelation when in the presence of free ion competing ligands, and we offer a theoretical framework to describe the coupled dynamic equilibria that result in this effect. We believe the insights presented here can be generally applied to advance engineering of the broadening class of polymer materials with dynamic cross-links.
Cazzell, Holten-Andersen, Expanding the stoichiometric window for metal cross-linked gel assembly using competition, PNAS, 2019.
Collaborations
Professor James Gilchrist Lehigh University
Professor Matt Harrington McGill University
Professor Martin Lenz LPTMS of CNRS and Université Paris-Sud
Professor Joseph Tracey North Carolina State University
Copyright © 2020-26 Holten-Andersen Group - All Rights Reserved.
